
A bright yellow precipitate of lead nitrate forms.Because it is very insoluble it is non-toxic, unlike other, soluble, barium compounds. Barium sulfate is used as a barium meal in medicine since it is opaque to X-rays. A dense white precipitate of barium sulfate forms.The following notes explain the expected observations for each step/reaction: Add a small piece of zinc metal to the copper sulfate solution. Put two drops of copper(II) sulfate solution into box 10.Observe closely using a magnifying glass. Add one drop of iron(II) sulfate to the silver nitrate solution. Put one drop of silver nitrate solution into box 9.Observe, and record any changes over the next ten minutes. 
Add two drops of sodium hydroxide to the barium nitrate solution. Put two drops of barium nitrate solution into box 8.Add two drops of iron(II) sulfate solution to the potassium manganate(VII) solution. Put two drops of potassium manganate(VII) solution into box 7.Add two drops of sodium hydroxide solution to the iron(II) sulfate solution. Put two drops of iron(II) sulfate solution into box 6.Add one drop of hydrochloric acid, then a small piece of zinc metal to the ammonium vanadate(V) solution. Put two drops of ammonium vanadate(V) solution into box 5.Add two drops of ammonia solution to the copper(II) sulfate solution. Put two drops of copper(II) sulfate solution into box 4.Add one drop of potassium thiocyanate solution to the iron(III) nitrate solution. Put two drops of iron(III) nitrate solution into box 3.Add two drops of potassium iodide solution to the drops of lead nitrate solution. Put two drops of lead nitrate solution into box 2.Add two drops of sodium sulfate solution to the drops of barium nitrate solution. Put two drops of barium nitrate solution into box 1 (at the top of the middle column).Zinc metal granules – see CLEAPSS Hazcard HC107.Ĭover the table on your worksheet with a clear plastic sheet, then follow steps 1–10 below.Potassium thiocyanate, KSCN(aq), 0.2 mol dm –3 – see CLEAPSS Hazcard HC095A and CLEAPSS Recipe Book RB122.Potassium manganate(VII), 0.01 mol dm –3 – see CLEAPSS Hazcard HC081 and CLEAPSS Recipe Book RB073.Iron(III) nitrate, Fe(NO 3) 3.9H 2O(aq), 0.2 mol dm –3 – see CLEAPSS Hazcard HC055C and CLEAPSS Recipe Book RB052.Iron(II) sulfate, FeSO 4.7H 2O(aq), 0.2 mol dm –3 – see CLEAPSS Hazcard HC055B and CLEAPSS Recipe Book RB051.Hydrochloric acid, HCl(aq), 1 mol dm –3 – see CLEAPSS Hazcard HC047a and CLEAPSS Recipe Book RB043.Sodium sulfate, Na 2SO 3(aq), 0.5 mol dm –3 – see CLEAPSS Hazcard HC098B and CLEAPSS Recipe Book RB107.Barium nitrate, Ba(NO 3) 2(aq), 0.2 mol dm –3 – see CLEAPSS Hazcard HC011 and CLEAPSS Recipe Book RB010.The following chemicals are all of low hazard:. 
See CLEAPSS Hazcard HC027c and CLEAPSS Recipe Book RB031. Copper(II) sulfate solution, CuSO 4(aq), 0.2 mol dm –3 causes eye damage and is HAZARDOUS to the aquatic environment.See CLEAPSS Hazcard HC091a and CLEAPSS Recipe Book RB085. Sodium hydroxide solution, NaOH(aq), 1 mol dm –3 is corrosive.Ammonium vanadate(V), NH 4VO 3, 0.2 mol dm –3 (acidified with sulfuric acid) is a mutagen and extremely toxic if inhaled – but not by any other route.See CLEAPSS Hazcard HC006 and CLEAPSS Recipe Book RB006. Ammonia solution, NH 3(aq), 3 mol dm –3 is CORROSIVE.See CLEAPSS Hazcard HC057a and CLEAPSS Recipe Book RB053. Lead nitrate, Pb(NO 3) 2(aq), 0.5 mol dm –3 is a reproductive toxin, causes eye damage, causes damage to organs (especially the CNS) and is harmful to the aquatic environment. 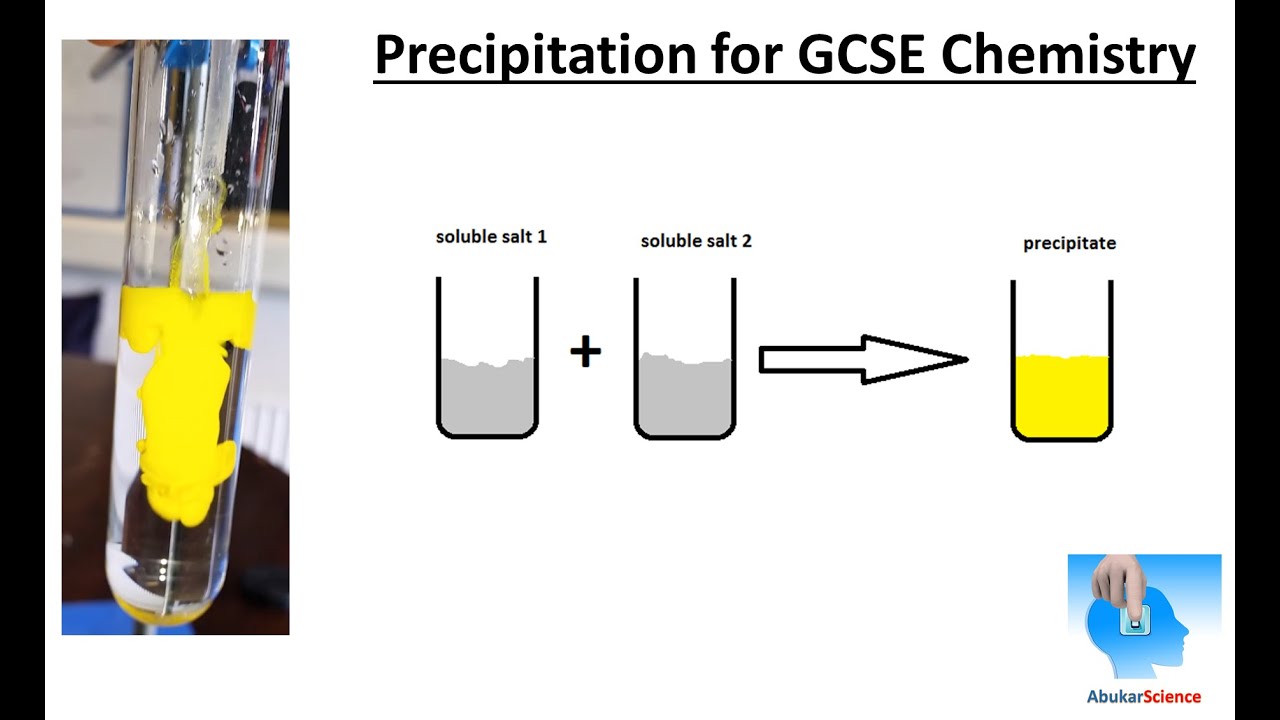
See CLEAPSS Hazcard HC087 and CLEAPSS Recipe Book RB077. Keep separate from organic waste containers.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
